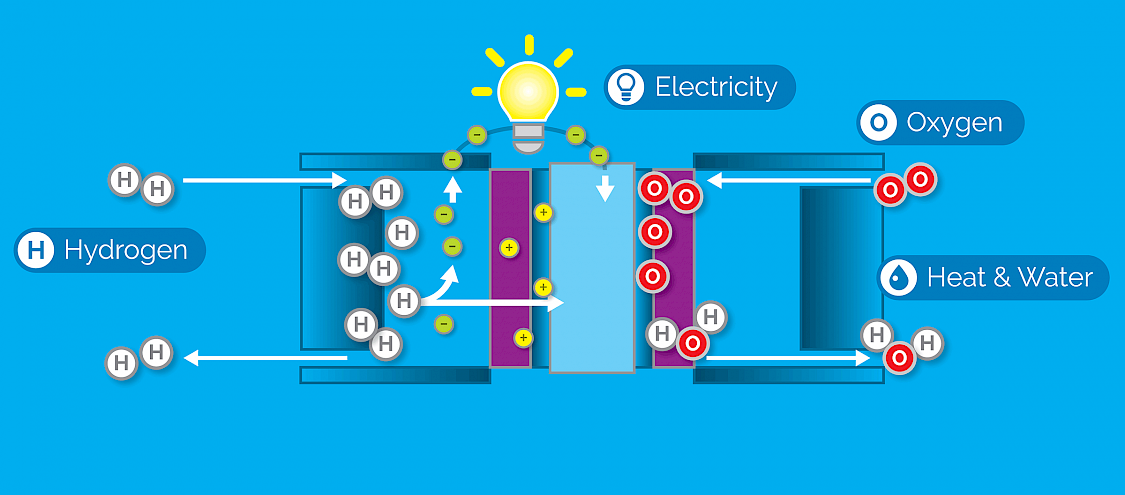
The Fuel Cell Process
- Hydrogen from the storage tank is pumped into one side of the fuel cell stack.
- Oxygen from the air enters the other side of the fuel cell stack.
- As the hydrogen naturally gravitates toward the oxygen, it passes through a membrane, separating the hydrogen molecules into electrons and protons.
- The protons move through the fuel stack while the electrons follow an external circuit, delivering electricity to an electric motor.
- When the electrons return to the fuel cell, they combine with the oxygen to produce clean water and heat – the only byproducts of the process.
Hydrogen is an extensively tested and proven fuel for everyday use. With a decades-long history of safe and effective use in the chemical and aerospace industries, hydrogen is just as safe as the gasoline used to fuel everyday vehicles. The lightest of all the elements, hydrogen is non-toxic and dissipates quickly into the atmosphere.
Vehicle Safety
Like all vehicles on the road, fuel cell electric vehicles (FCEVs) are extensively tested and designed with safety in mind by major auto manufacturers all over the world. Vehicles are outfitted with a number of standard safety systems and features including carbon fiber-wrapped on-board fuel storage tanks, which must meet applicable DOT criteria before being used in a vehicle. As further proof of their safety, hydrogen fuel cell forklifts are being widely used today in large warehouses. There is probably a hydrogen vehicle in use close to where you work, live, or commute.
Codes and Standards
Hydrogen codes and standards regulate the safe production, manufacturing, use and maintenance of hydrogen fuel, vehicles and refueling stations. For an up-to-date directory of all relevant codes and standards, check out the Hydrogen and Fuel Cells Codes and Standards Matrix.
Want to Learn More?
Clean
Compressed hydrogen in and pure water out. That's the simplicity of a fuel cell electric vehicle (FCEV). With zero greenhouse gases or air pollution emitted during operation, FCEVs are paving the way for a future of sustainable transportation.
Efficient
A hydrogen fuel cell is two to three times as efficient as an internal combustion engine, so FCEVs can easily store the fuel needed to travel 300 miles before refueling.
Refuels Quickly
Unlike battery electric vehicles, which can take hours to charge, FCEVs fill up in minutes using familiar technology similar to standard gasoline and diesel filling stations.
Quiet
With no mechanical gears or combustion, FCEVs only make a quiet hum, even at highway speeds.
Safe
Hydrogen is an extensively tested and proven fuel that is just as safe as the gasoline used to power everyday vehicles. Vehicles are outfitted with a number of standard safety systems and features, which must meet applicable DOT criteria before hitting the road.
Proven
FCEVs have racked up millions of test miles and are proven reliable and ready for use anywhere in the world. Most manufacturers have a fuel cell vehicle that is ready to go now or will be very soon. In fact, a new hydrogen roadmap released by the Renewable Hydrogen Fuel Cell Collaborative and Stark Area Regional Transit Authority lays out a plan of action to deploy 135,000 fuel cell electric vehicles, build 250 hydrogen stations and create 65,000 new jobs over next 15 years.
Want to Learn More?
- Hydrogen Roadmap for the U.S. Midwest Region
- How Do Fuel Cell Electric Vehicles Work? (Alternative Fuels Data Center)
- Compare Fuel Cell Vehicles (Office of Transportation & Air Quality)
- Refueling (RHFCC)
By making the switch to hydrogen fuel, transit agencies are doing their part to conserve natural resources, improve the health in our cities and create a greener, cleaner future for our citizens without compromising on vehicle performance. Individual fuel cells can be assembled into stacks to increase power, making them a scalable solution for full-size passenger buses and smaller paratransit applications.
Clean
With clean water as the only emission, hydrogen-powered buses release zero pollutants or greenhouse gases into the air, putting the health of our people and planet first.
Efficient
Fuel cell electric-powered buses are 2-3 times more efficient than those with internal combustion engines and will eliminate the consumption of 9,000 gallons of fuel over the lifetime of the vehicle.
Safe
Hydrogen is a non-toxic, extensively tested and proven fuel that is just as safe as the gasoline used to power everyday vehicles.
Flexible
Hydrogen fuel cell buses fill up in minutes and are the most flexible option for zero-emission transportation. Like diesel buses, they operate on a long-duty cycle of 12-16 hours without re-fueling.
Quiet
With only a quiet hum, hydrogen-powered buses are more enjoyable for the riders and reduce noise pollution in the cities they serve.
Proven Success
More than 80 fuel cell buses are in operation or planned around the country, including in Ohio, Michigan, California, South Carolina, Texas, Delaware, Hawaii, Alabama and New York. Hundreds of fuel cell buses are also running routes through Europe and Asia. In many countries, the fuel cell bus has become the standard for mass transit because the buses can be used interchangeably with diesel buses, but are zero emission and completely quiet. Read more program success stories.
Ready to Go Hydrogen?
Learn more about how the Midwest Hydrogen Center of Excellence can help connect your transit agency with transformative funding opportunities.
Want to Learn More?
Hydrogen fuel cell-powered lift trucks are being put to work in warehouses all over the country. With more than 21,000 operating in at least 26 states and a two-year return on investment over and above battery-powered units, hydrogen fuel cell-powered lift trucks are a smart and economic option for materials handling. In the state of Ohio alone, there are more than 1,000 fuel cell lift trucks in operation.
Zero Emissions
Unlike propane-powered lift trucks, hydrogen fuel cell-powered lift trucks emit zero greenhouse gases or harmful pollutants.
Long Range
Hydrogen fuel cell-powered lift trucks run up to three times longer than battery-powered units and eliminate the need for battery changes during a shift.
Consistent Power
Unlike battery-powered units, which can vary in performance throughout the last half of the battery charge, hydrogen fuel cell-powered units deliver consistent power until the fuel tank is depleted.
Fast Fueling
Fueling takes minutes and can be performed at an on-site generation system operated by lift truck operators.
Economic
Fuel cell lift trucks enjoy very attractive economics as compared to electric vehicles. Fuel cells last longer than battery packs and do not require a complex recharging management plan or warehouse space for recharging racks.
Midwest Deployments
- Wal-Mart
- Amazon
- Honda
- Home Depot
- Ace Hardware
- Kroger Co.
- Central Grocers
- Golden State Foods
- Sysco
- Testa Produce
Want to Learn More?
Hydrogen fuel cell electric vehicles (FCEVs) are completely zero-emission and can be easily scaled to meet the needs of diverse applications. This makes them a viable option for transportation applications including light-duty, medium-duty delivery and heavy-duty freight trucks and municipal and commercial fleets, which may include any number of vehicle types.
FCEVs can be fueled by locally generated renewable alternative fuels, making them an attractive option for municipal, commercial and delivery fleets looking for green vehicle options. Because fleet vehicles typically return to home base after completing their mission each day, municipalities and private fleets are good candidates for on-site refueling stations. Once operating, these stations can also serve the public by providing fuel for passenger FCEVs.
Current interest is focused on a hybrid approach to large vehicles – a battery electric base with a fuel cell range extender. Examples on the road today include:
- Paratransit vehicle - Based on the battery electric Nissan eNV 200, this vehicle has 6 kg of hydrogen storage and a 5 kW fuel cell, extending the range from 80 to 250 miles.
- Nikola tractor - Made for over-the-road trucking, the 1,000 hp truck has a range of up to 1,200 miles.
- Toyota Class-8 Trucks - "Toyota and Kenworth will deploy a total of 10 trucks as part of the Zero and Near-Zero Emissions Freight Facilities Project (ZANZEFF), hauling cargo received at the Ports of Los Angeles and Long Beach, throughout the LA Basin."
Want to Learn More?
Production
Hydrogen is most often found in a compound with other elements (H2O) and must be isolated in order to power fuel cell electric vehicles (FCEVs). While it is not an energy source itself, it is an excellent carrier of energy and can be produced domestically from a diverse array of resources including fossil fuels, biomass and water electrolysis with electricity.
The majority of hydrogen today is produced by natural gas reforming, a cost-effective process that most commonly employs steam-methane reforming to isolate hydrogen. These large centrally located reforming units are already transporting liquid and compressed gas hydrogen to various locations for industrial applications.
Hydrogen can also be produced on-site and with renewable energy through methods including:
- Electrolysis
- Using biogas from municipal sludge and food wastes to power a high temperature fuel cell, which produces hydrogen directly
- Using a semiconductor material to produce hydrogen directly from sunlight
Fueling
Unlike battery electric vehicles, which can take hours to charge, FCEVs fill up in minutes using familiar technology similar to standard gasoline and diesel stations. Hydrogen is dispensed as a compressed gas and can power a vehicle for 300 miles before refueling.
The Renewable Hydrogen Fuel Cell Collaborative is committed to researching, developing and implementing renewable hydrogen-based infrastructure as rapidly as possible. In the next six years, the RHFCC expects to implement 50 stations throughout the Midwest region with 10 percent of these being renewable-based.